

Effortlessly Manage Your Electronic Submissions Assessment
An all-in-one end-to-end submission process for regulatory agencies
Efficiently validate submissions by predefining requirements
Ensure that medicinal products meet regulatory requirements
Scale your processes with parallel work between multiple users
How much time do you spend assessing submissions?
Reviewing large volumes of data for compliance takes a lot of time without dedicated tools and processes.
- I’m pressured to reduce reviewing time and comply with procedural timelines
- We’re overwhelmed by the number of submissions to process
- I’m having difficulties identifying the changes in a submission within its lifecycle
- I’m frustrated by the lack of collaboration because documents are stored in multiple data silos
- We’re struggling with a complex infrastructure and are in need of a smarter automation

Validating and reviewing submissions within given timelines should be effortless
Efficient handling
Reporting
Validation at scale
Lifecycle management
"EXTEDO is a great example of a company that really understands the requirements of the market."
Technical Operations and International Business at Genericon
Your plan to effortless compliance
Schedule a call
Get a free consultation
Reduce your assessment timelines
Streamline your submission validation


Request your personal EXTEDO Software Demo now!
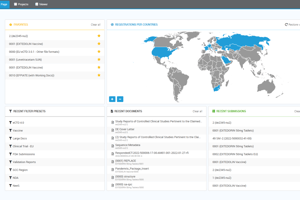
Easy to deploy, swift to scale
The agency solution is powerful, yet simple to deploy within your agency. Depending on your system specifications, the agency solution can be installed and validated within a matter of days. Its user-friendly and intuitive interface is equipped with integrated, context-sensitive online help that ensures ease of adoption amongst your users and minimizes the need for expensive structured training sessions. Supporting distributed workforces with many hundreds of users, the agency solution is an extremely scalable solution.
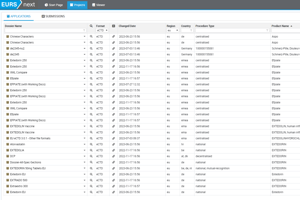
Quick access to relevant information by text search and filtering
Through the agency solution interface, you can access imported submissions and search, view, and annotate them. This powerful feature allows you to organize and filter submissions based on certain criteria, such as regulatory activities corresponding to the overall regulatory status of a drug product. As recognized by regulatory authorities around the globe, the agency solution is the requisite solution for effortless review and validation of regulatory submissions.
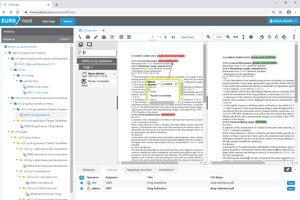
The efficient user interface
If you need online access to previously submitted electronic dossiers, then EXTEDO’s agency solution is the answer. It provides a fully functional reviewing system for agencies, enabling them to view and review submission data directly within a web browser. With no need for client software, the agency solution is the ideal solution for collaborative working between agency staff and external assessors where IT resources may be limited.
Integrates effortlessly with third-party software
Some of our Customers




Services

-
Business Process and Regulatory Consulting Services
Tailored specifically to the needs of regulatory and related stakeholders, EXTEDO’s business process and regulatory consulting services are designed to support you during and after your eCTD submissions. Through a series of workshops, our team of experienced consultants will establish your business needs, understand your processes, and help you to define the most appropriate implementation approach.
-
Education & Training Services
To ensure you get the most out of your purchased solution, we offer detailed training for each product within the EXTEDOpulse solution portfolio. Training sessions are tailored to your individual needs and cover a broad range of technical and regulatory topics. Designed to educate you on how to utilize your EXTEDO solution, our training sessions are conducted either in-house or onsite.
-
Technical Consulting
Purchasing a new EXTEDO application is the first step to streamlining business and regulatory processes within your organization. However, ensuring correct installation, implementation and integration is a crucial step in the process of deploying your new solution.


