
EXTEDOpulse Submission Publishing – Effortlessly Manage, Publish and Validate Regulatory Submissions
Improve submission quality and consistency
Validate and publish submissions from a single solution
How many tools do you need to manage your submissions?
- I am overwhelmed by the number of places where data is stored
- We lack the processes, expertise, and infrastructure to scale up
- The day doesn’t have enough hours to complete my regular tasks
- I struggle with the increasing number and complexity of submissions I need to compile
- We are struggling to find a solution that fits into our IT infrastructure
- We do have a very low user acceptance because the current software is not user-friendly and error-prone
- Our processes struggle to keep up with regulatory requirements

Complying with regulatory standards and processes around the world should be effortless.
Dossier assembly
Handles all global standards
Submission validation
Lifecycle management
Keep track of all changes and updates to your submissions.
"I have been using EXTEDO’s submission publishing and validation solution since the inception of eCTD, and EXTEDO has consistently provided impeccable guidance throughout the many changes in this field over the years."
Your plan to effortless compliance
Schedule a call
We’ll discuss your goals and uncover your challenges with viewing and reviewing electronic submissions.
Get a free consultation
Ensure effortless submission compliance with global regulations
Streamline your submission management
Creating and maintaining regulatory submissions that are compliant with regulatory standards around the globe is complex and time-consuming.
From authoring and publishing to ongoing management, EXTEDOpulse streamlines end-to-end regulatory submission processes, enabling organizations to scale their operations and generate error-free, compliant submissions.


Request your personal EXTEDO Software Demo now!
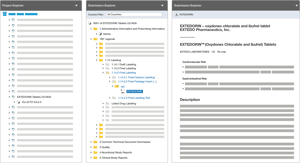
The most comprehensive eCTD management software solution available
With the EXTEDOpulse Submission Management Hub, you are able to compile, view, validate and publish compliant submissions based on eCTD v3, eCTD v4, NeeS, ACTD, eCopy, IMPD, PIP, VNeeS, DMF, ASMF, Clinical Trial Applications and other regional formats. EXTEDOpulse significantly improves the quality and consistency of your submissions, with a powerful hyperlinking and bookmarking engine that allows the detection, notification and correction of broken links. For FDA electronic submissions, EXTEDOpulse provides sophisticated Structured Product Labelling (SPL) capabilities that also enable data entry and maintenance of product information.
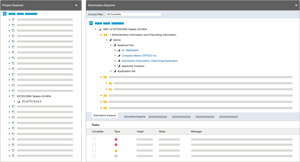
An integrated solution that simplifies your lifecycle management
EXTEDOpulse’s Submission Management Hub goes beyond basic eCTD submissions software. It provides you with a complete regulatory dossier assembly environment that enables your organization to operate in a compliant manner within a heavily regulated environment. Easily handle electronic submissions without prior knowledge of the regulatory standard. The in-built technical validation ensures your submission is valid according to the latest validation criteria provided by the authorities.
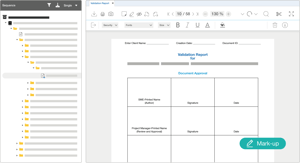
Save time and costs for validation and compliance
Through our shared validation approach, the EXTEDOcloud reduces the time and cost involved in validating your EXTEDOpulse Submission Publishing instance. Within your cloud environment, EXTEDO ensures that your solution is Installation Qualification (IQ), Operational Qualification (OQ), and Performance Qualification (PQ) validated. Where required, we can provide validation scripts and documentation for your records. The resulting system is validated under FDA 21 CFR Part 11 and EU GMP Annex 11 Volume 4.
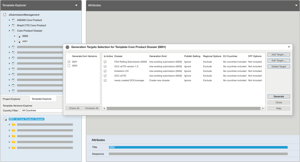
Streamline and maintain global submissions with the parent-child dossier
Through the use of a parent-child dossier concept, DOCmanager, as an add-on to EXTEDOpulse Submission Publishing, enables you to readily generate responsive templates for multiple submissions through simple country-level changes. Any changes you make to the parent dossier are automatically inherited by the child dossier, thus eliminating the need for revalidation. Reuse the content for dossiers based on different submission scenarios, thus reducing the efforts associated with compiling and maintaining multi-region product dossiers.
Export and submit clinical studies in any eSubmission management tool with the RLPmanager module
The RLPmanager module add-on for report-level publishing gives you the power to compile and export files into a single format eligible for submissions in EXTEDOpulse Submission Management Hub or any other electronic submission management software. Use drag-and-drop to create, compile, validate and version clinical studies, or edit them at a later date. When you’re ready, you can merge multiple PDF files into a single document, ready for use in any electronic submission.

Guided Transition to Publish Compliant eCTD v4 Submissions
Transitioning from eCTD v3 to eCTD v4 can be complex — but you don’t have to navigate this evolution alone. Our Submission Management Hub guides you through every step of the publishing process, ensuring your applications are fully compliant with the latest regional and technical requirements. From handling new metadata structures to managing lifecycle changes more efficiently, we provide the tools and expertise to simplify the shift. Convenient access to controlled vocabulary and forward compatibility automation, makes the transition easier than ever imagined.
Some of our Customers




.png?width=150&height=70&name=Vieco%20Pharmaceutical%20(FZCO).png)
Services

Business Process and Regulatory Consulting Services
Education & Training Services
Technical Consulting
Available Modules

EXTEDO DOCmanager Module

EXTEDO RLPmanager Module
Other Resources






