

Simplify your authoring process of eCTD dossiers with eCTDtemplates
Retain consistency, cut down review times and optimize dossier production.
Designed and developed around industry regulations and best practices
Regularly updated to ensure compliance with the latest authoring guidance
Cut down review times with consistent layouts and comprehensive instructional guidelines
Creating documents for electronic submissions can be a time-consuming, inconsistent process involving many different authors, working with numerous different files.
eCTDtemplates provide a simple way to create consistent and compliant dossiers in eCTD (electronic Common Technical Document) and other formats.
All eCTD dossiers start their life as a set of individual documents, authored around common structures and containing standard information. With each individual author writing in their own style, the resulting documents are often inconsistent in appearance and format. This often leads to a lengthy review, both internally and at the agencies.
Through their library of over 1300 pre-written Microsoft Word templates, eCTDtemplates deliver a common starting point for all your technical documentation. By ensuring consistent authoring standards, it enables authors to focus on content and automate the style. This reduces common errors and provides a harmonized structure for non-regulatory personnel.

Request your personal EXTEDO Software Demo now!
Reduce re-work and speed up approvals
Inconsistent writing styles and non-compliant information within documentation can lead to time-consuming updates and rewrites. At best, this could result in approvals being delayed by the authorities or worse, it can result in agencies refusing to accept files during their technical assessment.
Through the provision of inline guidance text for authors, eCTDtemplates indicate what information needs to be entered into each individual area for each document type. EXTEDO’s eCTDtemplates also ensure that your information is structured consistently and accurately entered. This enables reviewers to quickly locate the information they need and makes process reviews and approvals faster.
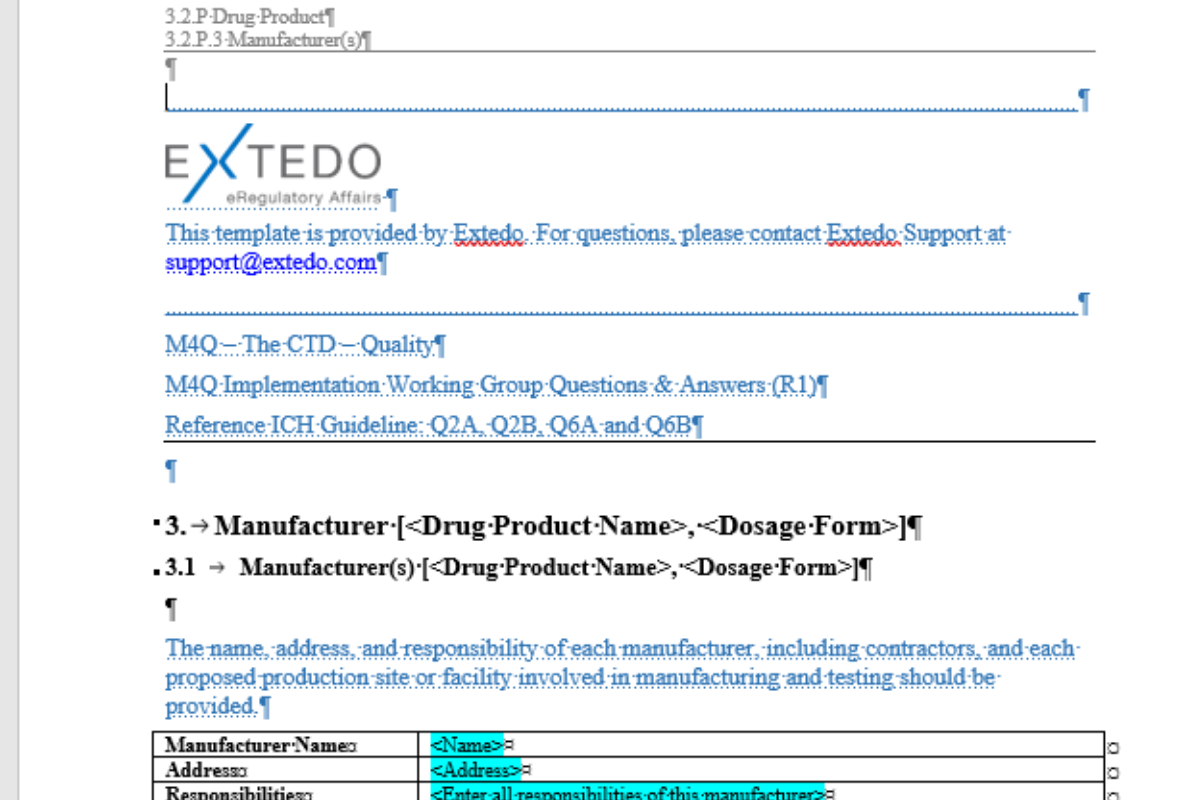
Speed up authoring for both Medical Writing and Regulatory Affairs
eCTDtemplates include the EXTEDO Word Toolbar to assist your authors in creating their content. The EXTEDO Word Toolbar contains 60 essential buttons to reduce the time needed to perform the most common authoring tasks in a single click. It can automatically create a Table of Content, Table of Figures or Table of Tables; add footnotes to a table, and remove your hidden text.
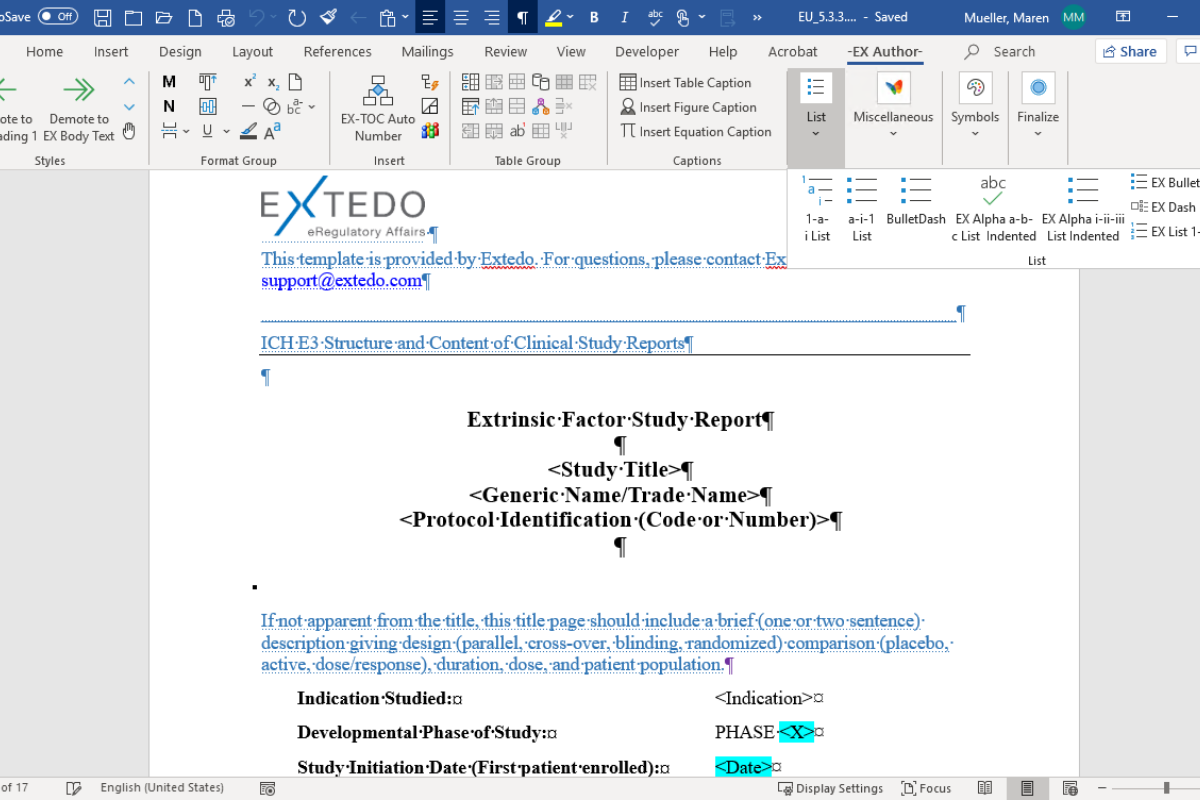
Effortless compliance in all your writing
eCTDtemplates provide Effortless Compliance™ with industry best practices and writing guidelines for eCTD documentation. They improve your documentation standards, minimize re-work and optimize time-critical document production processes. This feature helps you remain compliant with every document you produce.

Get your eCTD submission ready, faster with automated hyperlinking and auto-compilation
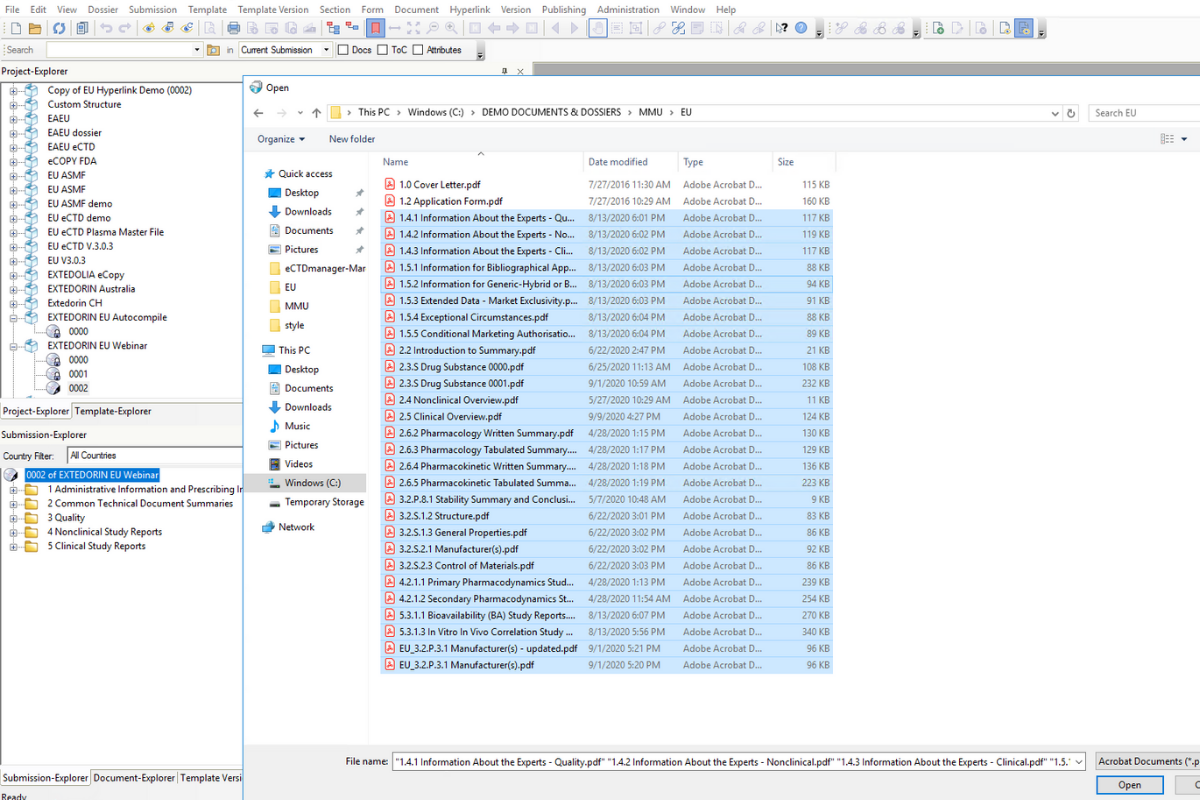
EXTEDO eCTDtemplates also make it easier for regulatory departments to make documents submission-ready. References can automatically be created as hyperlinks when the document is converted to PDF. Similarly, the use of heading styles allows for the automatic creation of bookmarks. And last, but not least, eCTDtemplates are prepared for auto-compilation in eCTDmanager. Now, you can import multiple documents into the submission and they will be positioned in the correct sections of the dossier, automatically.


“EXTEDO is a great example of a company that really understands the requirements of the market.”
Director Business Development, Technical Operations and international Business at Genericon
Your plan to effortless compliance
Schedule a call
We’ll discuss your goals and uncover your challenges with your document authoring process.
Get a free consultation
Our experienced business and technical team will outline a solution to solve your challenges.
Simplify your authoring process of eCTD dossiers
Retain consistency, cut down review times and optimize dossier production.
Some of our Customers
.png)




Book a meeting
Education & Training Services
To ensure you get the most out of your purchased solution, we offer detailed training for each product within the EXTEDOpulse solution portfolio. Training sessions are tailored to your individual needs and cover a broad range of technical and regulatory topics. Designed to educate you on how to utilize your EXTEDO solution, our training sessions are conducted either in-house or onsite.
Technical Consulting
Purchasing a new EXTEDO application is the first step to streamlining business and regulatory processes within your organization. However, ensuring correct installation, implementation and integration is a crucial step in the process of deploying your new solution.

